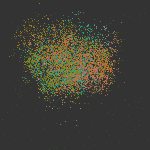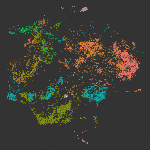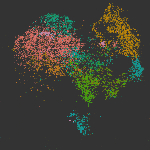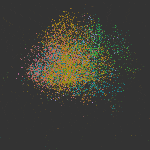
The cellular and molecular changes that occur during the initiation and progression of TAAD are much less studied compared with AAAD, especially for the large number of sporadic cases that are more affected by environmental factors rather than genetic predisposition. In this study, we performed single-cell RNAs-seq of thoracic aortic cells from BAPN (β-aminopropionitrile)-induce TAAD mouse models at three time points that span the early and advanced stages of the disease. Comparative analyses of single-cell data between BAPN-induce TAAD models and healthy controls were performed to identify the alterations in cellular composition, lineage-specific regulation and cell-cell communication during the development of TAAD, especially at the early stage. Unbiased clustering of 61,826 cells revealed 24 clusters, which were assigned to 7 cell lineages. Differential proportional analysis uncovered SMC and macrophage subpopulations that were associated with the progression of TAAD. Pathways that were dysregulated in SMCs during the development of TAAD were identified through GSEA, which highlights the contribution of oxidative stress, ER stress and activation of Toll like receptor signaling pathways to SMC senescence at the early stage of TAAD. Furthermore, potentially key molecules of SMCs at the early stage of TAAD were found through gene regulatory network analysis, which highlights the role of stress-responsive genes expressed in SMCs at the initiation stage. Cell-cell communication analysis revealed enhanced intercellular communications in TAAD, and further found that macrophages may contribute to TNF-mediated apoptosis and activation of TGF beta signaling in SMCs at the early stage. In addition, we molecularly characterized the three subpopulations of aortic macrophages that were significantly expanded during the development of TAAD: Lyve1+ resident-like macrophages (cluster c10), Cd74high antigen presenting macrophages (cluster c14) and Il1rn+ tissue-infiltrated proinflammatory macrophages (cluster c13). The presence of the three macrophage subpopulations was then validated in both mouse and human aortic tissue via smFISH. Based on the single-cell data together with bulk RNA-seq data of samples from human patients, we demonstrated that in both mice and humans, aortic tissue specific macrophages, especially the Il1rn+ proinflammatory macrophage subpopulation, represent the predominant source of most detrimental molecules that contribute to the development of TAAD, such as MMPs and inflammatory cytokines. Finally, we show that depletion of the aortic macrophages with an inhibitor Ki20227 could mitigate the progression and decrease the incidence of TAAD in mice.
Fuwai Hospital
Founded in 1956, Fuwai Hospital which belongs to the Chinese Academy of Medical Sciences (referred as “Fuwai Hospital”) is a national triple A, first-class hospital specializing in cardiovascular diseases. It is also theNational Center for Cardiovascular Diseases, State Key Laboratory of Cardiovascular Disease, and National Clinical Research Center of Cardiovascular Diseases.It is well known internationally for treatment of all kinds of complex, difficult, and severe cardiovascular diseases, and has become the world’s largest cardiovascular center, as well as a national center for clinical healthcare, medical research, disease prevention and education.

Contact Us

Xuanyu Liu
Bioinformatics,
Molecular genetics, Machine learning,
Understanding development and disease with single-cell approaches
swissox2017@sina.com

Wen Chen
Molecular Genetics,
Next-Generation Sequencing,
Understanding development and disease with single-cell approaches
chen-cheery@foxmail.com

Wenke Li
Bioinformatics,
Algorithm, Software,
Artificial Intelligence
Database, Web Development, Data Visualization
wk1lian@126.com